oncoFISH her2
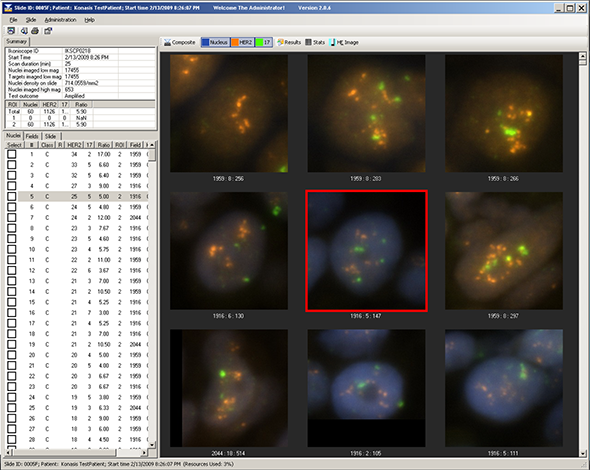
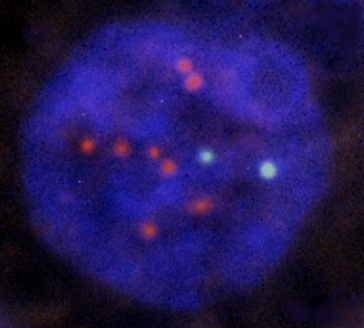
Amplification of the human epidermal growth factor receptor type 2 gene (HER2) is associated with adverse clinical outcomes in patients with both lymph node negative and lymph node positive breast cancer. FDA Cleared HER2 amplification is associated with enhanced response rates to trastuzumab therapy, a monoclonal antibody that targets the extracellular portion of the HER2 receptor, and both taxane and anthracycline-based chemotherapy regimens. oncoFISH her2 is a fully automated microscopy application for the determination of the HER2 status of tissue sections from breast tissue biopsies, that have been hybridized with a FISH probe for the HER2 gene and a control probe for the chromosome 17 centromere. The area of the tissue section that contains invasive carcinoma is usually marked by the pathologist on the H&E slide. An image of the H&E slide is displayed in combination with the DAPI image of the FISH preparation. The pathologist or cytotechnologist can then mark one or more area(s) of interest to be scanned for determination of HER2 status. The entire delineated area is scanned using a 20x objective for initial FISH signal analysis. Select areas were then imaged using a 100x objective to determine the number of FISH signals per cell and the Her2:CEP17 ratio. The system provides a testing algorithm for accurate, reproducible assay performance and the ability to provide an image of the H&E slide and to match the H&E and FISH slides helps ensure that invasive tumor is used for reporting the assay. In addition to consistency, an automated system has the benefit of providing a record of all the images produced for a case. Such a record can be of immediate benefit in allowing multiple reviewers of a difficult or equivocal case, without the risk of bleaching of the signals and long term benefit in terms of providing a permanent record of a case.